WVE Posts Interim Data From Phase I Study on WVE-007, Stock Tanks
Shares of Wave Life Sciences WVE plunged 49.6% yesterday after the company announced new data from the phase I portion of the INLIGHT study evaluating its investigational pipeline candidate, WVE-007, in otherwise healthy individuals who are overweight or living with obesity.
What Did WVE’s Latest Data Show?
Data from this study showed that after six months of follow-up, a single 240 mg dose of WVE-007 demonstrated sustained benefits compared to placebo, including a statistically significant 14% reduction in visceral fat and a 5% reduction in total body fat. Lean mass remained stable with a 2% increase, while additional improvements were observed in waist circumference, which decreased by 3%, and overall body weight, which declined by 1%.
However, in the 400 mg group, three-month results showed a 5% reduction in visceral fat and a 0.7% decline in total fat versus placebo following a single dose of WVE-007. Lean mass remained largely unchanged, with a negligible 0.2% decrease, suggesting preservation of muscle, though overall fat reduction was relatively modest at this interim stage.
This might have hurt investors' sentiments and resulted in the stock declining following the announcement of the news.
Treatment with WVE-007 led to greater improvement in body composition, as measured by the visceral fat-to-muscle ratio, at six months compared with weekly GLP-1 therapies evaluated in later-stage studies.
Treatment with WVE-007 was generally safe and well-tolerated to date, up to 600 mg.
Year to date, shares of Wave Life SciencesWVE-- have plunged 63.5% against the industry’s rise of 1.6%.
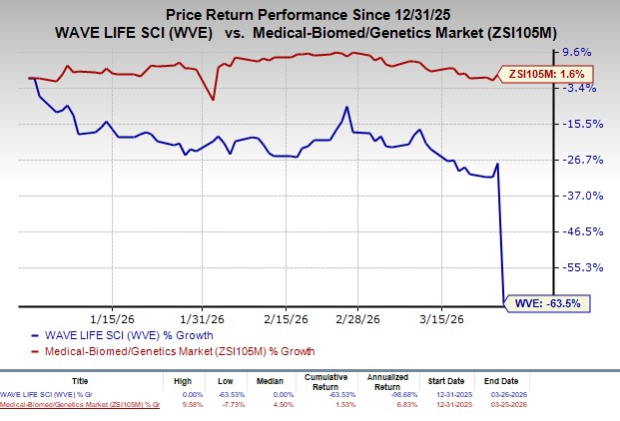
Image Source: Zacks Investment Research
WVE Other Development Activities With WVE-007
The company is gearing up to initiate the phase IIa multidose portion of the INLIGHT study in the second quarter of 2026. This part of the study will evaluate WVE-007 as a monotherapy in individuals with higher BMI (35–50 kg/m²) and associated comorbidities.
Data from this study will inform further development of WVE-007 in obesity as well as metabolic dysfunction-associated steatohepatitis (MASH), type II diabetes, and cardiovascular disease.
Additional data from the INLIGHT study, including results from the 600 mg of WVE-007 phase I single ascending dose (SAD) cohort, are expected later in 2026.
The company also plans to initiate a new clinical study evaluating WVE-007 as an incretin add-on and as post-incretin maintenance later in 2026.
Stiff Competition in the Obesity Space
The obesity market has garnered significant attention over the past few years due to the sizable and still underpenetrated market opportunity. Currently, Eli Lilly LLY and Novo Nordisk NVO dominate this space with their respective injectable therapies.
Eli Lilly and Novo Nordisk are trying to gain an upper hand in oral weight-loss pills. In late December, the FDA approved an oral version of NVO’s Wegovy. The drug was commercially launched in early January. The approval made Wegovy the first GLP-1 therapy available as a pill for weight management, offering a needle-free alternative that could expand patient adoption. A regulatory filing seeking approval for Eli Lilly’s obesity pill orforglipron is currently under review by the FDA, with a final decision expected soon.
Several other companies, like Viking Therapeutics VKTX, are also making rapid progress in the obesity space. Last year, VKTX started two late-stage studies evaluating the subcutaneous formulation of its investigational obesity drug, VK2735. While one of these studies completed enrolment at a rapid pace, VKTX recently completed enrolment in the other study, too.
WVE’s Zacks Rank
Wave Life Sciences currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Just Released: Zacks Top 10 Stocks for 2026
Hurry – you can still get in early on our 10 top tickers for 2026. Handpicked by Zacks Director of Research Sheraz Mian, this portfolio has been stunningly and consistently successful.
From inception in 2012 through November, 2025, the Zacks Top 10 Stocks gained +2,530.8%, more than QUADRUPLING the S&P 500’s +570.3%.
Sheraz has combed through 4,400 companies covered by the Zacks Rank and handpicked the best 10 to buy and hold in 2026. You can still be among the first to see these just-released stocks with enormous potential.
See New Top 10 Stocks >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novo Nordisk A/S (NVO): Free Stock Analysis Report
Eli Lilly and Company (LLY): Free Stock Analysis Report
Viking Therapeutics, Inc. (VKTX): Free Stock Analysis Report
WAVE Life Sciences Ltd. (WVE): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet