Regeneron Wins FDA Nod for Extended Eylea HD Dosing Interval
Regeneron Pharmaceuticals REGN announced that the FDA has approved extending dosing intervals of up to every 20 weeks for Eylea HD (aflibercept) in patients with wet age-related macular degeneration (wAMD) and diabetic macular edema (DME) who have demonstrated a sustained response after one year.
The approval is based on 96-week data from two pivotal studies (PULSAR and PHOTON) showing the majority of Eylea Hd patients maintained their visual and anatomic improvements with extended dosing intervals.
As part of the approval, the regulatory body updated Eylea HD’s label to incorporate 96-week (two-year) data from the pivotal PULSAR trial in wAMD and PHOTON trial in DME, confirming sustained efficacy and safety with extended dosing intervals over two years.
With this approval, treatment can now be individualized — ranging from as frequent as every four weeks to as infrequent as every 20 weeks — based on patient response, offering greater flexibility while maintaining outcomes.
Per REGNREGN--, the new Eylea HD dosing regimen enables patients with wAMD and DME to receive treatment as infrequently as two to three times per year, further expanding the broadest dosing interval range among approved injectable anti-VEGF therapies.
More on Regeneron’s Eylea HD
Regeneron developed Eylea HD to deliver efficacy and safety comparable to its lead drug, Eylea, but with fewer injections.
Eylea, an anti-VEGF therapy approved across multiple ophthalmology indications, remains Regeneron’s largest revenue contributor. However, declining Eylea sales continue to pressure the company’s overall revenues.
Competitive pressure has intensified, particularly from Roche’s RHHBY Vabysmo, which targets both angiopoietin-2 (Ang-2) and VEGF-A pathways and has gained meaningful traction in the retinal disease market.
RHHBY Vabysmo’s sales grew 12% to CHF 4.1 billion in 2025 on strong demand across all regions.
While lead drug Eylea continues to face headwinds, Eylea HD is gaining traction, supported by steady label expansions.
Eylea HD sales rose 36% in 2025 to $1.6 billion in the United States.
In November 2025, the FDA approved Eylea HD for the treatment of macular edema following retinal vein occlusion (RVO), with dosing of up to once every eight weeks after an initial monthly dosing phase. The FDA also approved a monthly dosing option for certain patients who may benefit from resuming this dosing schedule across all currently approved indications, including wAMD, DME, diabetic retinopathy and RVO.
Eylea and Eylea HD were co-developed with Bayer BAYRY.
Regeneron records net product sales in the United States, while Bayer records sales outside the country.
The FDA has also set an April 2026 target action date for the Eylea HD prefilled syringe, which is currently under review via a Chemistry, Manufacturing and Controls Prior-Approval Supplement.
REGN’s Efforts to Strengthen Portfolio Beyond Eylea Franchise
The going has been strong for RegeneronREGN-- over the past six months. Shares of this biotech giant have surged 30.6% in this time frame, outpacing the industry’s growth of 13.1%.
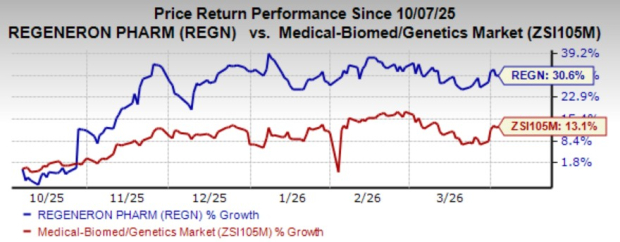
Image Source: Zacks Investment Research
REGN’s top line also comprises its share of profits/losses in connection with the global sales of Dupixent. Partner Sanofi SNY records global net product sales of Dupixent.
Strong demand across approved indications — including atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis and eosinophilic esophagitis — continues to drive robust growth. Ongoing label expansions for Dupixent remain a key contributor to revenue visibility and profitability.
Regeneron’s oncology franchise is anchored by its PD-1 inhibitor Libtayo (cemiplimab-rwlc), which is approved for use in certain patients with advanced basal cell carcinoma, advanced cutaneous squamous cell carcinoma and advanced non-small cell lung cancer.
Libtayo delivered a solid performance in 2025, generating $1.4 billion in sales, which reflects a 19% year-over-year increase. Regeneron records global net product sales of Libtayo and pays royalties to partner Sanofi on these sales.
Progress across its oncology portfolio should support greater diversification of revenues and reduce reliance on any single franchise.
REGN currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Radical New Technology Could Hand Investors Huge Gains
Quantum Computing is the next technological revolution, and it could be even more advanced than AI.
While some believed the technology was years away, it is already present and moving fast. Large hyperscalers, such as Microsoft, Google, Amazon, Oracle, and even Meta and Tesla, are scrambling to integrate quantum computing into their infrastructure.
Senior Stock Strategist Kevin Cook reveals 7 carefully selected stocks poised to dominate the quantum computing landscape in his report, Beyond AI: The Quantum Leap in Computing Power.
Kevin was among the early experts who recognized NVIDIA's enormous potential back in 2016. Now, he has keyed in on what could be "the next big thing" in quantum computing supremacy. Today, you have a rare chance to position your portfolio at the forefront of this opportunity.
See Top Quantum Stocks Now >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Regeneron Pharmaceuticals, Inc. (REGN): Free Stock Analysis Report
Sanofi (SNY): Free Stock Analysis Report
Roche Holding AG (RHHBY): Free Stock Analysis Report
Bayer Aktiengesellschaft (BAYRY): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.



Comments
No comments yet