MRNA Stock Gains After CHMP Endorses COVID-19-Influenza Combo Shot
Shares of Moderna MRNA rose about 4% on Friday after the company announced that the EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended granting marketing authorization to mCombriax (mRNA-1083), its mRNA-based combination vaccine against COVID-19 and influenza.
The CHMP’s recommendation will now be reviewed by the European Commission and a final decision is expected shortly. If approved, mCombriax would be the company’s fourth marketed product in the European Union, after two COVID-19 vaccines, Spikevax and mNexspike, and RSV vaccine mResvia.
This recommendation was supported by data from a pivotal phase III study, which met its primary endpoints. Data from the study showed that a single dose of mCombriax elicited higher immune responses against COVID-19 and influenza compared with licensed standalone COVID and influenza vaccines.
These results are based on a study that evaluated the vaccine in about 8,000 adults, divided equally into two independent age group cohorts. The first cohort includes adults aged 65 and older and compares mCombriax with the co-administration of Fluzone HD, a high-dose influenza vaccine marketed by Sanofi SNY, and Spikevax. The second group consists of adults aged 50 to 64, comparing the combination vaccine with Fluarix, a standard-dose influenza vaccine marketed by GSK plc GSK, co-administered with Spikevax.
MRNA Stock Price Performance
Year to date, the stock has surged about 82% compared with the industry’s 10% growth.
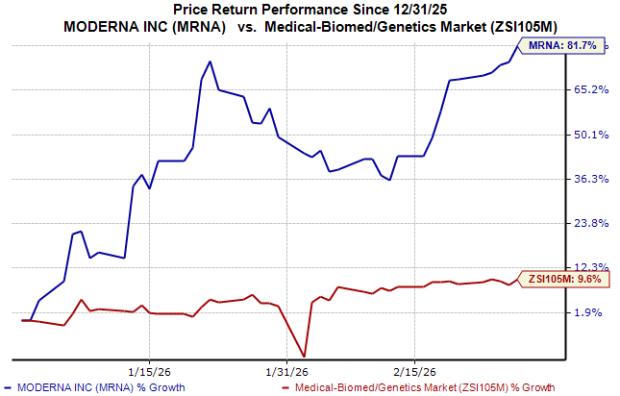
Image Source: Zacks Investment Research
More on Moderna’s mCombriax
mCombriax combines Moderna’s two vaccines, mNexspike and mRNA-1010, an investigational influenza vaccine. Regulatory filings are currently under review seeking approval in the United States, Europe, Canada and Australia. A final decision by the FDA is expected by Aug. 5, 2026.
Moderna had initially submitted a regulatory filing for mCombriax with the FDA. This filing was voluntarily withdrawn in May 2025 after the agency requested additional efficacy data for the flu component. The requested data has been submitted as one of the studies supporting the mRNA-1010 filing. The company is currently awaiting further guidance from the FDA for a potential filing of mCombriax.
The heightened level of scrutiny against vaccines in the United States has been weighing on overall vaccine demand. Initially, the FDA refused to review the regulatory filing for mRNA-1010. Per the agency, the late-stage study supporting the filing was not adequate and well-controlled because the licensed standard-dose seasonal influenza vaccine that the company used as the comparator was not “the best-available standard of care.”
The FDA reversed its decision after Moderna proposed a regulatory pathway for mRNA-1010’s approval, based on age. The regulatory filing was subsequently modified, which now seeks full approval for the vaccine in adults aged 50-64 years and an accelerated approval for adults aged 65 years and above.
MRNA’s Zacks Rank
Moderna currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
5 Stocks Set to Double
Each was handpicked by a Zacks expert as the favorite stock to gain +100% or more in the months ahead. They include
Stock #1: A Disruptive Force with Notable Growth and Resilience
Stock #2: Bullish Signs Signaling to Buy the Dip
Stock #3: One of the Most Compelling Investments in the Market
Stock #4: Leader In a Red-Hot Industry Poised for Growth
Stock #5: Modern Omni-Channel Platform Coiled to Spring
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor. While not all picks can be winners, previous recommendations have soared +171%, +209% and +232%.
See Our Newest 5 Stocks Set to Double Picks >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Sanofi (SNY): Free Stock Analysis Report
GSK PLC Sponsored ADR (GSK): Free Stock Analysis Report
Moderna, Inc. (MRNA): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet