LLY's Next-Gen Obesity Drug Meets Goal in First Phase III Study for T2D
Eli Lilly LLY announced positive top-line results from a late-stage study evaluating its experimental once-weekly weight-loss drug, retatrutide, in adults with type II diabetes (T2D)
This study, called TRANSCEND-T2D-1, evaluated three doses (4 mg, 9 mg and 12 mg) of the drug for 40 weeks against placebo in adults with T2D and inadequate glycemic control with diet and exercise alone. Eli LillyLLY-- reported results using two approaches — an efficacy estimand, which reflects outcomes in patients who stayed on treatment throughout the study period and a treatment-regimen estimand, which incorporates data from patients who discontinued treatment.
The study achieved its primary endpoint. Under the efficacy estimand, patients who took retatrutide achieved A1C reduction (a measure of blood sugar levels) by an average of 1.7% to 2% compared to 0.8% in the placebo group. Across the treatment-regimen estimand, patients receiving the drug reduced A1C by an average of 1.7% to 1.9%, versus 0.8% in the placebo group.
For a key secondary goal, patients who took the drug lost up to 16.8% of their body weight using the efficacy estimand, compared to 2.5% in the placebo group. Under the treatment-regimen estimand, patients lost 15.3% of their body weight, compared to 2.6% in the placebo group. LLYLLY-- noted that the weight loss continued through the end of the treatment period.
The TRANSCEND-T2D-1 is the second of the eight late-stage studies evaluating retatrutide across various cardiometabolic indications, including T2D, obesity with at least one weight-related medical problem, sleep apnea and chronic low back pain. In December, Eli Lilly reported top-line data from the late-stage TRIUMPH-4 study, which evaluated the drug in adults with obesity or overweight and knee osteoarthritis, and without diabetes. Results from this study showed that retatrutide lowered weight by up to an average of 28.7%. Data from some of the remaining studies are expected later this year.
Retatrutide activates three hormone receptors — GLP-1, GIP and glucagon — compared with the company’s approved obesity drug Zepbound, which targets GLP-1 and GIP. In comparison, Novo Nordisk’s NVO obesity drug Wegovy activates only GLP-1.
Eli Lilly’s Stock Movement
As seen in the chart below, Eli Lilly’s shares have underperformed the industry year to date.
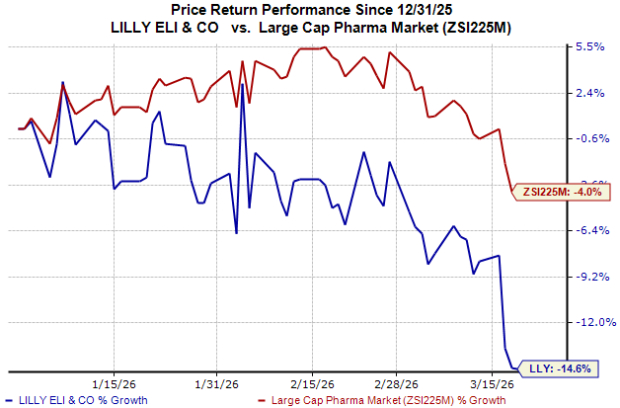
Image Source: Zacks Investment Research
Competition Escalates in the Obesity Space
The obesity market has garnered significant attention over the past few years due to the sizeable and still underpenetrated market opportunity. Currently, Eli Lilly and Novo Nordisk dominate this space with their respective injectable therapies.
Eli Lilly and Novo Nordisk are racing to introduce oral weight-loss pills. In late December, the FDA approved an oral version of Wegovy. The drug was commercially launched in early January. The approval made Wegovy the first GLP-1 therapy available as a pill for weight management, offering a needle-free alternative that could expand patient adoption. A regulatory filing for Eli Lilly’s obesity pill orforglipron is currently under review by the FDA, with a final decision expected soon.
Several other companies, like Viking Therapeutics VKTX, are making rapid progress in the obesity space. Last year, VKTX started two late-stage studies evaluating the subcutaneous formulation of its investigational obesity drug, VK2735. While one of these studies recently completed enrolment at a rapid pace, the company expects to complete enrolment in the other study soon.
Last year, Pfizer PFE closed the acquisition of obesity drug developer Metsera for around $10 billion, after a heated bidding war against Novo Nordisk. The Metsera acquisition has brought Pfizer back into the lucrative obesity space by adding the latter’s four novel clinical-stage incretin and amylin programs, which are expected to generate billions of dollars in peak sales. In December, Pfizer announced that it is in-licensing exclusive global rights to develop and market an oral small molecule GLP-1 drug from a leading Chinese drugmaker.
LLY’s Zacks Rank
Eli Lilly currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
#1 Semiconductor Stock to Buy (Not NVDA)
The incredible demand for data is fueling the market's next digital gold rush. As data centers continue to be built and constantly upgraded, the companies that provide the hardware for these behemoths will become the NVIDIAs of tomorrow.
One under-the-radar chipmaker is uniquely positioned to take advantage of the next growth stage of this market. It specializes in semiconductor products that titans like NVIDIA don't build. It's just beginning to enter the spotlight, which is exactly where you want to be.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Pfizer Inc. (PFE): Free Stock Analysis Report
Novo Nordisk A/S (NVO): Free Stock Analysis Report
Eli Lilly and Company (LLY): Free Stock Analysis Report
Viking Therapeutics, Inc. (VKTX): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet