Will the Recent Label Expansion of BMY's Sotyktu Boost Its Prospects?
Bristol Myers BMY recently secured a meaningful label expansion for Sotyktu, with the FDA approving the drug for adults with active psoriatic arthritis (PsA). The decision adds a second major indication beyond plaque psoriasis, strengthening the product’s long-term commercial profile and lifecycle potential.
From an investment standpoint, this approval introduces a differentiated, oral therapy into a large and competitive PsA market historically dominated by injectable biologics.
Sotyktu’s position as the first TYK2 inhibitor approved for PsA could support uptake, particularly among patients and physicians seeking convenient, non-biologic options earlier in the treatment paradigm.
Strategically, the label expansion reinforces Bristol Myers’ immunology franchise and diversifies revenue streams beyond oncology. With psoriatic arthritis affecting a meaningful subset of psoriasis patients, Sotyktu now addresses both skin and joint manifestations, increasing its addressable market and cross-selling potential within existing dermatology and rheumatology channels.
However, commercial execution will be key. The PsA market is highly competitive, with entrenched biologics and oral agents already available.
Overall, the PsA approval represents a catalyst for BMYBMY--, enhancing Sotyktu’s growth outlook and supporting a broader shift toward immunology as a durable revenue pillar.
BMY is working on the label expansion of its drugs to strengthen its growth portfolio, which primarily comprises Opdivo, Orencia, Yervoy, Reblozyl, Opdualag, Abecma, Zeposia, Breyanzi, Camzyos, Sotyku, Krazati and others.
The drug raked in sales of $291 million in 2025, up 19% from 2024. Sotyktu is also being evaluated for lupus (data is expected later in the year) and Sjogren’s Disease.
Schizophrenia drug Cobenfy is being evaluated for psychosis associated with Alzheimer's disease, for agitation in Alzheimer's Disease and Alzheimer's Disease cognition.
The successful development of all late-stage pipeline candidates and label expansion of approved drugs should be a significant boost for BMY as its legacy portfolio continues to be adversely impacted by the continued generic impact on Revlimid, Pomalyst, Sprycel and Abraxane.
Competition for BMY’s Sotyktu
Sotyktu faces stiff competition from Amgen’s AMGN Otezla in the psoriasis space.
Amgen acquired global commercial rights to Otezla from erstwhile Celgene (now part of Bristol-Myers). Amgen is also evaluating Otezla in additional indications.
Novartis’ NVS blockbuster drug Cosentyx is approved for various conditions like hidradenitis suppurativa, psoriatic arthritis, plaque psoriasis, ankylosing spondylitis, and non-radiographic axial spondyloarthritis, as well as several pediatric inflammatory disorders.
Cosentyx is a fully human monoclonal antibody that blocks interleukin-17A, a cytokine involved in inflammatory pathways underlying several immune-mediated diseases.
It is one of the top drugs for NVS. The drug generated $6.7 billion in revenues for NVS in 2025, representing 8% growth from 2024.
BMY’s Price Performance, Valuation & Estimates
Shares of Bristol MyersBMY-- have lost 4.8% over the past year against the industry’s growth of 11.2%.
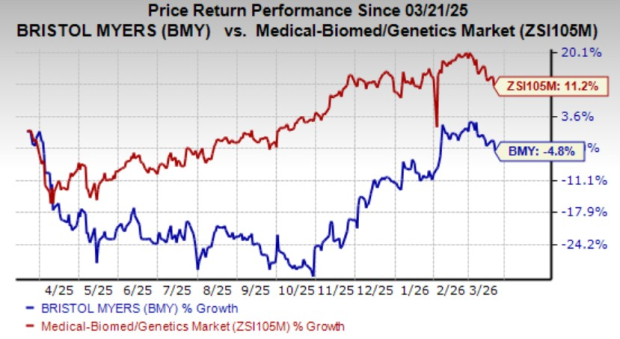
Image Source: Zacks Investment Research
From a valuation standpoint, BMY is trading at a discount to the large-cap pharma industry. Going by the price/earnings ratio, the stock currently trades at 9.34x forward earnings, higher than its mean of 8.48x but lower than the large-cap pharma industry’s 16.92x.
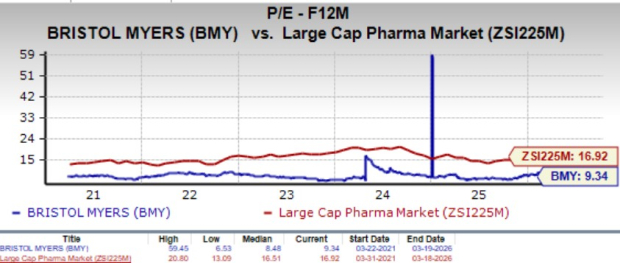
Image Source: Zacks Investment Research
The Zacks Consensus Estimate for 2026 EPS has moved north to $6.26 from $6.15 in the past 30 days, while that for 2027 has increased to $6.09 from $5.94.
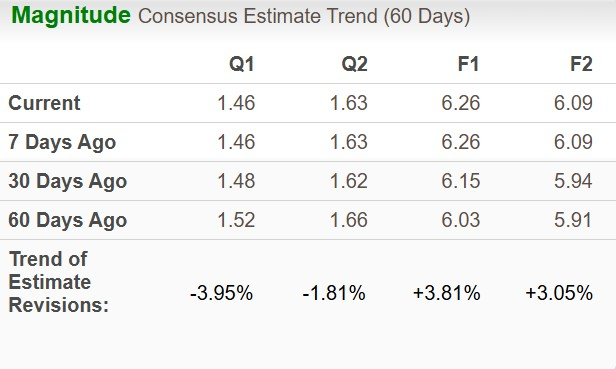
Image Source: Zacks Investment Research
BMY currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
#1 Semiconductor Stock to Buy (Not NVDA)
The incredible demand for data is fueling the market's next digital gold rush. As data centers continue to be built and constantly upgraded, the companies that provide the hardware for these behemoths will become the NVIDIAs of tomorrow.
One under-the-radar chipmaker is uniquely positioned to take advantage of the next growth stage of this market. It specializes in semiconductor products that titans like NVIDIA don't build. It's just beginning to enter the spotlight, which is exactly where you want to be.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS): Free Stock Analysis Report
Bristol Myers Squibb Company (BMY): Free Stock Analysis Report
Amgen Inc. (AMGN): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet