FDA Grants Priority Tag to Ionis' NDA for Alexander Disease Drug
Ionis Pharmaceuticals IONS announced that the FDA has granted priority review to its new drug application (NDA) seeking approval for zilganersen for the treatment of patients with Alexander disease (AxD), a rare, progressive and often fatal neurological disorder.
The regulatory body has set a target action date of Sept. 22, 2026.
Priority Review is granted to drugs that could significantly improve the treatment, prevention or diagnosis of serious conditions, enabling the FDA to review applications in about six months instead of the standard 10 months.
If approved, zilganersen, an RNA-targeted therapy, would become Ionis’ third wholly owned approved drug, joining Tryngolza (for familial chylomicronemia syndrome) and Dawnzera (for hereditary angioedema). It would also mark Ionis’ commercial entry into the neurology space.
AxD is a rare neurological disease that affects a type of brain cell called astrocytes, which play multiple roles in the brain to support neurons and oligodendrocytes. People living with this condition experience cognitive dysfunction and progressive neurologic deterioration, including difficulty swallowing and the inability to control muscles for large movements. There are currently no disease-modifying medicines approved for AxD patients.
Year to date, shares of IonisIONS-- have declined 10.5% compared with the industry’s 5.2% fall.
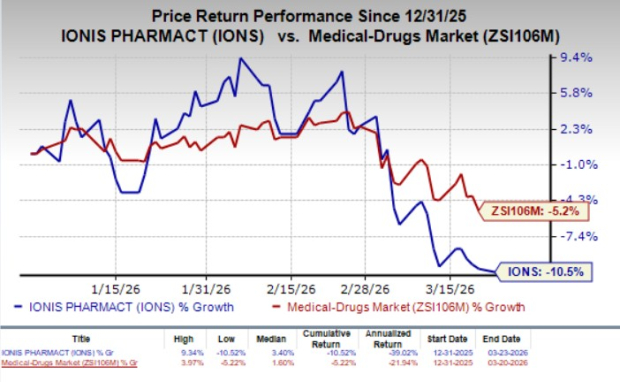
Image Source: Zacks Investment Research
Ionis Wins FDA Priority Review Backed by Strong Phase III Data
The FDA’s priority review was based on positive top-line data from a pivotal phase III study evaluating zilganersen in children and adults living with AxD. The study met its primary endpoint — patients who received a 50 mg dose of zilganersen achieved a statistically significant and clinically meaningful stabilization in gait speed compared with control. Treatment with the candidate also demonstrated a favorable safety and tolerability profile.
Though Ionis did not disclose whether key secondary endpoints were statistically significant, it reported that treatment with zilganersen showed a “consistent benefit” across these measures, indicating evidence of slowed disease progression, stabilization or improvement.
IONS’ Zacks Rank & Stocks to Consider
Ionis currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Some better-ranked stocks in the biotech sector are Catalyst Pharmaceuticals CPRX and Indivior Pharmaceuticals INDV, each currently sporting a Zacks Rank #1 (Strong Buy), and ANI Pharmaceuticals ANIP, which presently carries a Zacks Rank #2 (Buy).
Over the past 60 days, estimates for Catalyst Pharmaceuticals’ 2026 earnings per share have risen from $2.55 to $2.87. CPRX shares have lost 9.3% over the past year.
Catalyst Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 35.19%.
Over the past 60 days, estimates for Indivior Pharmaceuticals’ 2026 earnings per share have risen from $2.79 to $3.08. INDV shares have surged 206.8% over the past year.
Indivior Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 74.53%.
Over the past 60 days, estimates for ANI Pharmaceuticals’ earnings per share have increased from $8.28 to $8.99 for 2026. Over the past year, shares of ANIP have rallied 11.5%.
ANI Pharmaceuticals' earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Beyond Nvidia: AI's Second Wave Is Here
The AI revolution has already minted millionaires. But the stocks everyone knows about aren't likely to keep delivering the biggest profits. Little-known AI firms tackling the world's biggest problems may be more lucrative in the coming months and years.
SeeWant the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Catalyst Pharmaceuticals, Inc. (CPRX): Free Stock Analysis Report
ANI Pharmaceuticals, Inc. (ANIP): Free Stock Analysis Report
Ionis Pharmaceuticals, Inc. (IONS): Free Stock Analysis Report
Indivior Pharmaceuticals Inc. (INDV): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet