Is Bristol Myers' Deep Pipeline the Key to Future Growth?
Bristol Myers Squibb’s BMY boasts a deep and promising pipeline. Key pipeline candidates with multi-billion-dollar potential are milvexian (Oral factor XIa inhibitor), admilparant (LPA1 antagonist) pumitamig (PD-L1 x VEGF-A bispecific antibody) and iberdomide & mezigdomide (oral CELMoD protein degraders).
In 2026, BMYBMY-- expects to report top-line registrational data for six candidates – milvexian in both atrial fibrillation and secondary stroke prevention, admilparant in idiopathic pulmonary fibrosis, iberdomide in relapsed or refractory multiple myeloma, mezigdomide and arlo-cel in relapsed or refractory multiple myeloma and RYZ101 in second-line plus GEPNETs.
The company recently announced positive interim results from the late-stage SUCCESSOR-2 study, evaluating the efficacy and safety of mezigdomide in combination with carfilzomib and dexamethasone (MeziKd) versus carfilzomib and dexamethasone (Kd) in patients with relapsed or refractory multiple myeloma (RRMM).
The results mark an important milestone for the company’s next-generation CELMoD program. SUCCESSOR-2 represents the first positive phase III study for mezigdomide and the second successful phase III study for BMY’s CELMoD program, reinforcing confidence in the company’s targeted protein degradation platform.
The company, in partnership with Johnson & Johnson JNJ, is developing milvexian for atrial fibrillation and secondary stroke prevention. The successful development of milvexian provides an opportunity to redefine anticoagulant therapy for thrombotic diseases.
Admilparant is being evaluated for idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis. The candidate has the potential to redefine the standard of care as a differentiated treatment. Data from the registrational ALOFT-IPF study is expected later in the year.
BMY and partner BioNTech are evaluating pumitamig for numerous solid tumor types.
Pumitamig is a novel investigational bispecific antibody, combining two complementary, validated mechanisms in oncology into a single molecule. Pumitamig combines PD-L1 checkpoint inhibition aimed at restoring T cells’ ability to recognize and destroy tumor cells with the neutralization of VEGF-A. Initial data is expected later in the year.
BMY is also working on label expansion of its approved drugs. The FDA recently approved a label expansion of psoriasis drug, Sotyktu (deucravacitinib), an oral, selective tyrosine kinase 2 inhibitor.
The regulatory body approved the drug for the treatment of adults with active psoriatic arthritis. Sotyktu is also being evaluated for lupus (data is expected later in the year) and Sjogren’s Disease.
Schizophrenia drug Cobenfy is being evaluated for psychosis associated with Alzheimer's disease, for agitation in Alzheimer's Disease and Alzheimer's Disease cognition.
The successful development of all these pipeline assets and label expansion of approved drugs should be a significant boost for BMY as its legacy portfolio continues to be adversely impacted by the continued generic impact on Revlimid, Pomalyst, Sprycel and Abraxane.
Competition for BMY’s Key Drugs
BMY’s growth portfolio primarily comprises Opdivo, Orencia, Yervoy, Reblozyl, Opdualag, Abecma, Zeposia, Breyanzi, Camzyos, Sotyku, Krazati and others.
Oncology is a key therapeutic area of focus for Bristol MyersBMY--, which is developing and delivering transformational medicines in this space.
However, BMY faces competition from large pharma companies like Merck MRK and Pfizer PFE.
The immuno-oncology space is dominated by pharma giant MRK’s blockbuster drug Keytruda (pembrolizumab).
Keytruda is approved for several types of cancer and alone accounts for around 50% of MRK’s pharmaceutical sales. Merck is currently working on different strategies to drive the long-term growth of Keytruda.
Developing bispecific antibodies that target two proteins, namely PD-1 and VEGF, has lately been one of the lucrative areas in cancer treatment.
In 2024, Merck received an exclusive global license to develop, manufacture and commercialize LM-299, a novel investigational PD-1/VEGF bispecific antibody from LaNova. Merck’s oncology portfolio boasts a blockbuster PD-L1 inhibitor, Keytruda, and the company is looking to build a diversified oncology pipeline spanning differentiated mechanisms and multiple modalities.
Pfizer is one of the largest and most successful drugmakers in the field of oncology. It has an innovative oncology product portfolio of antibody-drug conjugates (ADCs), small molecules, bispecifics and other immunotherapies that treat a wide range of cancers, including certain types of breast cancer, genitourinary cancer and hematologic malignancies, as well as certain types of melanoma, gastrointestinal, gynecological and lung cancer. Pfizer also has oncology biosimilars in its portfolio.
PFE inked a licensing agreement with 3SBio for the development, manufacturing and commercialization of SSGJ-707, a bispecific antibody targeting PD-1 and VEGF, outside China.
BMY’s Price Performance, Valuation & Estimates
Shares of Bristol Myers have lost 0.5% over the past year against the industry’s growth of 16.6%.
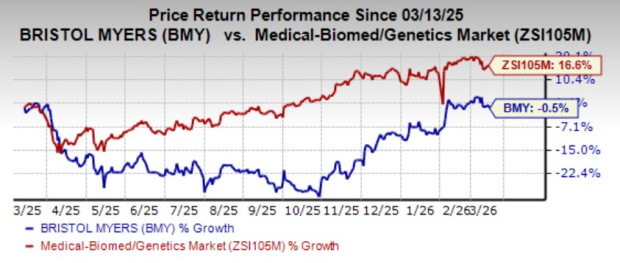
Image Source: Zacks Investment Research
From a valuation standpoint, BMY is trading at a discount to the large-cap pharma industry. Going by the price/earnings ratio, the stock currently trades at 9.64x forward earnings, higher than its mean of 8.47x but lower than the large-cap pharma industry’s 17.99x.

Image Source: Zacks Investment Research
The Zacks Consensus Estimate for 2026 EPS has moved north to $6.26 from $6.15 in the past 30 days, while that for 2027 has increased to $6.09 from $5.94.

Image Source: Zacks Investment Research
BMY currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks Names #1 Semiconductor Stock
This under-the-radar company specializes in semiconductor products that titans like NVIDIA don't build. It's uniquely positioned to take advantage of the next growth stage of this market. And it's just beginning to enter the spotlight, which is exactly where you want to be.
With strong earnings growth and an expanding customer base, it's positioned to feed the rampant demand for Artificial Intelligence, Machine Learning, and Internet of Things. Global semiconductor manufacturing is projected to explode from $452 billion in 2021 to $971 billion by 2028.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Bristol Myers Squibb Company (BMY): Free Stock Analysis Report
Johnson & Johnson (JNJ): Free Stock Analysis Report
Pfizer Inc. (PFE): Free Stock Analysis Report
Merck & Co., Inc. (MRK): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.

Comments
No comments yet