Bayer Wins EC Approval to Expand Kerendia in Heart Failure Treatment
Bayer BAYRY announced that the European Commission ("EC") has approved a label expansion of Kerendia (finerenone) in the European Union ("EU").
The drug, a selective, non-steroidal mineralocorticoid receptor antagonist (nsMRA), is now approved in the EU for the treatment of adults with heart failure (HF) with left ventricular ejection fraction (LVEF) ≥40%, i.e. HF with mildly reduced (HFmrEF) or preserved LVEF (HFpEF).
We remind investors finerenone is already marketed as Kerendia—or as Firialta in certain countries—and is approved in more than 100 countries, including China, Europe, Japan, and the United States, for the treatment of adults with chronic kidney disease (CKD) associated with type II diabetes (T2D).
Bayer Aktiengesellschaft Price, Consensus and EPS Surprise
Bayer Aktiengesellschaft price-consensus-eps-surprise-chart | Bayer Aktiengesellschaft Quote
More on BAYRY’s Kerendia’s Latest Label Expansion
The EC’s decision is based on results from the phase III FINEARTS-HF study, which demonstrated that finerenone significantly reduced the composite endpoint of cardiovascular death and total (first and recurrent) heart failure events, including hospitalizations and urgent visits, compared with placebo on top of standard therapy.
Heart failure affects more than 64 million people globally, including at least 15 million in Europe. Roughly half of these patients have LVEF ≥40%, a group often burdened by comorbidities such as CKD, hypertension, and atrial fibrillation, all of which contribute to increased hospitalizations and mortality. Despite the growing prevalence, treatment options remain limited, and this population is projected to represent the majority of heart failure hospitalizations in the near future.
The study is part of the ongoing MOONRAKER program — one of the largest phase III study initiatives in heart failure to date — enrolling more than 15,000 patients and designed to provide a comprehensive evaluation of finerenone across a wide range of patient populations and clinical settings.
The MOONRAKER program comprises the completed phase III FINEARTS-HF study, along with the ongoing investigator-sponsored collaborative studies REDEFINE-HF, CONFIRMATION-HF, and FINALITY-HF.
On the CKD side, the THUNDERBALL program comprises completed phase III studies — FIDELIO-DKD, FIGARO-DKD, FIND-CKD, and FINE-ONE — along with the phase II CONFIDENCE study and ongoing phase III pediatric studies FIONA and FIONA-OLE.
Earlier this month, finerenone achieved the primary endpoint in the late-stage FIND-CKD study, marking a key milestone in expanding its use to patients with non-diabetic CKD.
Kerendia is one of the top drugs for Bayer and generated sales of €829 million in 2025, up 79% from 2024.
BAYRY Looks to Strengthen Portfolio
Bayer’s recent turnaround has been phenomenal and the company is now looking to expand its diverse portfolio.
Shares of Bayer have surged 84.4% over the past year compared with the industry’s gain of 8.6%.
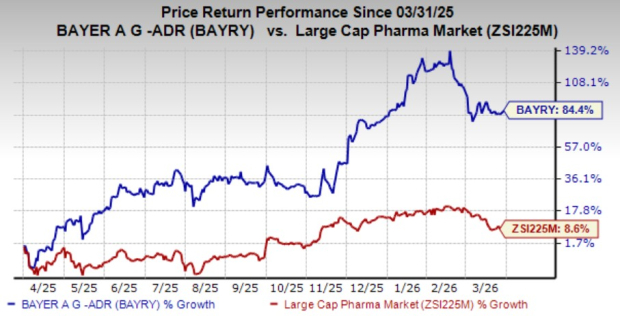
Image Source: Zacks Investment Research
Its new products, such as prostate cancer drugs Nubeqa and Kerendia, continue to maintain their impressive momentum in the Pharmaceutical division.
The strong performance of these drugs makes up for the decline in sales of oral anticoagulant Xarelto, which is co-developed with Johnson & Johnson JNJ.
Xarelto is marketed by Johnson & Johnson in the United States. Bayer earns license revenues from JNJ for Xarelto sales in the United States.
Eylea sales continue to face pressure from generics. The introduction of Eylea 8 mg, with its extended dosing intervals, has partially offset the decline and supported overall performance.
Please note that Bayer’s HealthCare unit co-develops Eylea with Regeneron REGN, which records net product sales of Eylea in the United States. BAYRY records net product sales of Eylea outside the country. REGN records its share of profits/losses in connection with the sales of Eylea outside the United States.
Label expansion of key drugs and approval of additional drugs will further boost sales from this business. The recent FDA approval of elinzanetant for the treatment of moderate to severe menopause-related vasomotor symptoms, under the brand name Lynkuet, is a significant boost for the company.
The FDA recently granted accelerated approval to Hyrnuo (sevabertinib) for the treatment of adult patients with locally advanced or metastatic non-squamous non-small cell lung cancer whose tumors have human epidermal growth factor receptor 2 tyrosine kinase domain-activating mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.
Encouraging developments in the Roundup litigation have lifted investor sentiment. Bayer acquired the Roundup weedkiller through its 2018 buyout of Monsanto. Since then, the company has faced numerous lawsuits alleging that Monsanto’s herbicide, whose active ingredient is glyphosate, caused cancer.
Monsanto has reached an agreement with leading plaintiffs’ law firms on a proposed class settlement related to the glyphosate litigation. This follows the U.S. Supreme Court’s decision to hear Bayer’s case, marking a crucial development for the company.
BAYRY expects litigation impacts to burden its cash position in 2026.
Zacks Rank
Bayer currently carries a Zacks Rank #5 (Strong Sell).
You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks' Research Chief Names "Stock Most Likely to Double"
Our team of experts has just released the 5 stocks with the greatest probability of gaining +100% or more in the coming months. Of those 5, Director of Research Sheraz Mian highlights the one stock set to climb highest.
This top pick is a little-known satellite-based communications firm. Space is projected to become a trillion dollar industry, and this company's customer base is growing fast. Analysts have forecasted a major revenue breakout in 2025. Of course, all our elite picks aren't winners but this one could far surpass earlier Zacks' Stocks Set to Double like Hims & Hers Health, which shot up +209%.
Free: See Our Top Stock And 4 Runners UpWant the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Regeneron Pharmaceuticals, Inc. (REGN): Free Stock Analysis Report
Johnson & Johnson (JNJ): Free Stock Analysis Report
Bayer Aktiengesellschaft (BAYRY): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
Zacks is the leading investment research firm focusing on equities earnings estimates and stock analysis for the individual investor, including stock picks, stock screening, portfolio stock tracker and stock screeners. Copyright 2006-2026 Zacks Equity Research, Inc. editor@zacks.com (Manaing editor) webmaster@zacks.com (Webmaster)
Latest Articles
Stay ahead of the market.
Get curated U.S. market news, insights and key dates delivered to your inbox.



Comments
No comments yet