CORT Rises on FDA Nod to Lifyorli in Platinum-Resistant Ovarian Cancer
Corcept Therapeutics CORT announced that the FDA has approved its selective glucocorticoid receptor antagonist, Lifyorli (relacorilant), in combination with nab-paclitaxel for the treatment of adult patients with platinum-resistant ovarian cancer.
The regulatory body has now approved Lifyorli in combination with nab-paclitaxel for treating adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens, at least one of which included bevacizumab.
Following the nod, Lifyorli has become the first FDA-approved selective glucocorticoid receptor antagonist for the given indication.
Shares of CorceptCORT-- were up 19.7% yesterday following the announcement of the news. The stock has rallied 16.3% so far this year against the industry’s decline of 4.9%.
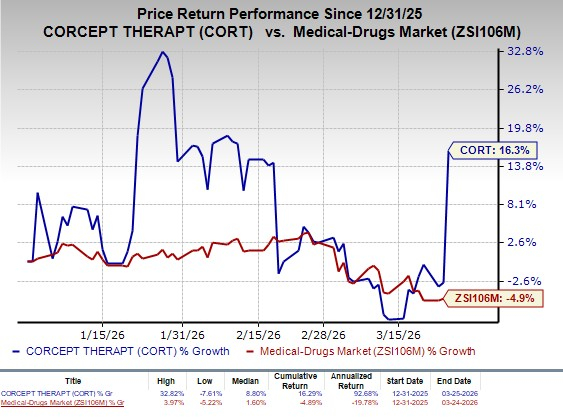
Image Source: Zacks Investment Research
How the FDA Nod for Lifyorli Helps CORT
The FDA approval for the Lifyorli combo was based on data from the pivotal phase III ROSELLA study. The study achieved its dual primary endpoints of progression-free and overall survival.
Data from the same showed that treatment with Lifyorli plus nab-paclitaxel led to a 35% reduction in the risk of death compared to nab-paclitaxel alone in the given patient population. Patients who were treated with Lifyorli plus nab-paclitaxel achieved a median OS of 16 months compared to 11.9 months in patients who received nab-paclitaxel alone.
Also, patients who were treated with Lifyorli in combination with nab-paclitaxel experienced a 30% reduction in the risk of disease progression versus patients who were treated with nab-paclitaxel alone. The combo of Lifyorli plus nab-paclitaxel was generally well tolerated.
Management believes that Lifyorli is now positioned to become a new standard-of-care treatment for platinum-resistant ovarian cancer.
The approval of Lifyorli is likely to provide a big boost to Corcept in diversifying its revenue base, given that the company’s top line was solely dependent on Korlym, which is approved for treating Cushing's syndrome. The approval of Lifyorli should lower the company’s heavy dependence on Korlym for revenues.
CORT’s Recent Development Activities With Relacorilant
Corcept is also developing relacorilant for treating Cushing's syndrome.
The company recently suffered a major setback when the FDA issued a complete response letter (“CRL”) to the new drug application (NDA) seeking approval for relacorilant to treat patients with hypercortisolism (Cushing’s syndrome). This NDA was based on positive data from the GRACE study, confirmatory evidence from the phase III GRADIENT study, long-term extension studies and a phase II study in hypercortisolism.
Corcept is currently engaging with the FDA to determine the best path forward to approval for relacorilant in Cushing’s syndrome.
Although for a different indication, the FDA approval of Lifyorli in platinum-resistant ovarian cancer is a positive development for Corcept, especially after it previously faced a setback in another indication.
Corcept has also submitted a marketing authorization application to the European Medicines Agency, seeking approval for relacorilant to treat patients with platinum-resistant ovarian cancer. A decision in Europe is expected later in 2026.
Relacorilant, in combination with other anticancer therapies, is also being studied in other solid tumors, including platinum-sensitive ovarian, endometrial, cervical and pancreatic cancers.
Corcept Therapeutics Incorporated Price
Corcept Therapeutics Incorporated price | Corcept Therapeutics Incorporated Quote
CORT’s Zacks Rank & Stocks to Consider
Corcept currently carries a Zacks Rank #4 (Sell).
Some better-ranked stocks in the drug/biotech sector are Avalo Therapeutics AVTX, ANI Pharmaceuticals ANIP and Replimune Group REPL, each carrying a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Over the past 60 days, estimates for Avalo Therapeutics’ 2026 loss per share have narrowed from $5.56 to $4.47, while the same for 2027 have narrowed from $2.60 to $2.27. AVTX’s shares have declined 23.2% year to date.
Avalo Therapeutics’ earnings beat estimates in one of the trailing four quarters and missed on the remaining three occasions, with the average negative surprise being 1.57%.
Over the past 60 days, estimates for ANI Pharmaceuticals’ 2026 earnings per share have increased from $8.28 to $8.99, while the same for 2027 have increased from $9.25 to $10.10. ANIP’s shares have fallen 6.2% year to date.
ANI Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Over the past 60 days, estimates for Replimune’s 2026 loss per share have narrowed from $3.61 to $3.40, while the same for 2027 have narrowed from $2.58 to $2.46. REPL’s shares have decreased 22.3% year to date.
Replimune’s earnings beat estimates in one of the trailing four quarters and missed on the remaining three occasions, with the average negative surprise being 4.46%.
Zacks Names #1 Semiconductor Stock
This under-the-radar company specializes in semiconductor products that titans like NVIDIA don't build. It's uniquely positioned to take advantage of the next growth stage of this market. And it's just beginning to enter the spotlight, which is exactly where you want to be.
With strong earnings growth and an expanding customer base, it's positioned to feed the rampant demand for Artificial Intelligence, Machine Learning, and Internet of Things. Global semiconductor manufacturing is projected to explode from $452 billion in 2021 to $971 billion by 2028.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Corcept Therapeutics Incorporated (CORT): Free Stock Analysis Report
ANI Pharmaceuticals, Inc. (ANIP): Free Stock Analysis Report
Replimune Group, Inc. (REPL): Free Stock Analysis Report
Avalo Therapeutics, Inc. (AVTX): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).

Comentarios
Aún no hay comentarios