COGT Rise as FDA Accepts Bezuclastinib NDA in Non-Advanced SM
Cogent Biosciences COGT announced that the FDA has accepted the new drug application (“NDA”) seeking approval for its lead pipeline candidate, bezuclastinib, for treating patients with non-advanced systemic mastocytosis (NonAdvSM).
With the FDA accepting the NDA for review, a decision from the regulatory body is expected on Dec. 30, 2026. The FDA also noted that no advisory committee is planned as of now and no review concerns have been identified.
Shares of Cogent BiosciencesCOGT-- were up 4.9% yesterday following the announcement of the news.
The stock has increased 0.5% so far this year compared with the industry’s rise of 2.8%.
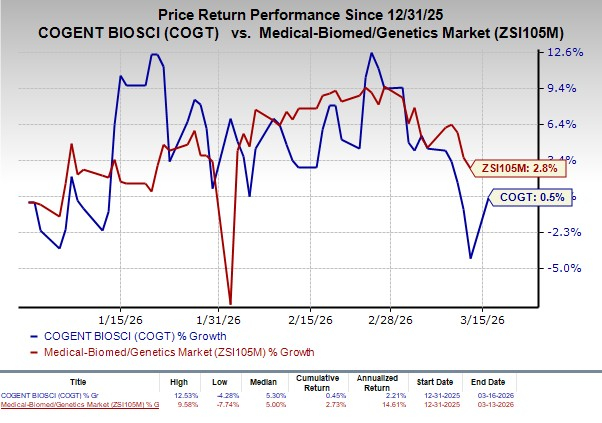
Image Source: Zacks Investment Research
COGT’s NDA for Bezuclastinib Based on SUMMIT Study
The above-mentioned NDA for bezuclastinib was based on data from the pivotal SUMMIT study.
The SUMMIT study achieved the primary and all key secondary endpoints, demonstrating clinically meaningful and highly statistically significant improvements following treatment with bezuclastinib in non-advanced SM patients.
Data from the SUMMIT study showed steadily deepening symptom improvement, suggesting lasting benefits following longer treatment with bezuclastinib. Patients who were treated with bezuclastinib did not just improve early on, but their symptoms kept getting better over time.
Treatment with bezuclastinib showed a favorable safety and tolerability profile, supporting its potential for chronic use in NonAdvSM, across the SUMMIT study.
COGT’s Other Development Activities With Bezuclastinib
The company is also evaluating bezuclastinib for another SM indication in a separate phase III study.
SM is a rare hematological disease impacting the immune system with debilitating symptoms that take a serious toll on the physical and psychological quality of life of patients.
The pivotal phase III APEX study is evaluating bezuclastinib for treating patients with advanced SM. The NDA for bezuclastinib in advanced SM is expected to be submitted in the first half of 2026.
Besides the SM indication, Cogent Biosciences is also developing bezuclastinib in combination with sunitinib for treating patients with gastrointestinal stromal tumors (“GIST”) who have received prior treatment with imatinib in the phase III PEAK study.
The NDA filing for the bezuclastinib combo for treating patients with second-line GIST is expected to be completed in April next month.
A potential approval for bezuclastinib is likely to give Cogent Biosciences the first commercial product and help it generate meaningful revenues.
COGT Zacks Rank & Stocks to Consider
Cogent Biosciences currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are Phathom Pharmaceuticals PHAT, ANI Pharmaceuticals ANIP and Replimune Group REPL, each carrying a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Over the past 60 days, estimates for Phathom Pharmaceuticals’ 2026 earnings per share have increased from 24 cents to 28 cents, while the same for 2027 have increased from $2.22 to $2.25. PHAT’s shares have plunged 37.1% in the year-to-date period.
Phathom Pharmaceuticals’ earnings beat estimates in two of the trailing four quarters, while missing the same on the remaining two occasions, with the average surprise being 14.06%.
Over the past 60 days, estimates for ANI Pharmaceuticals’ 2026 earnings per share have increased from $8.14 to $8.72, while the same for 2027 have increased from $9.25 to $10.10. ANIP’s shares have fallen 8.6% in the year-to-date period.
ANI Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Over the past 60 days, estimates for Replimune’s 2026 loss per share have narrowed from $3.59 to $3.41, while the same for 2027 have narrowed from $2.42 to $2.33. REPL’s shares have declined 24.3% in the year-to-date period.
Replimune’s earnings beat estimates in one of the trailing four quarters and missed on the remaining three occasions, with the average negative surprise being 4.46%.
5 Stocks Set to Double
Each was handpicked by a Zacks expert as the favorite stock to gain +100% or more in the months ahead. They include
Stock #1: A Disruptive Force with Notable Growth and Resilience
Stock #2: Bullish Signs Signaling to Buy the Dip
Stock #3: One of the Most Compelling Investments in the Market
Stock #4: Leader In a Red-Hot Industry Poised for Growth
Stock #5: Modern Omni-Channel Platform Coiled to Spring
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor. While not all picks can be winners, previous recommendations have soared +171%, +209% and +232%.
See Our Newest 5 Stocks Set to Double Picks >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
ANI Pharmaceuticals, Inc. (ANIP): Free Stock Analysis Report
Cogent Biosciences, Inc. (COGT): Free Stock Analysis Report
Replimune Group, Inc. (REPL): Free Stock Analysis Report
Phathom Pharmaceuticals, Inc. (PHAT): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).

Comentarios
Aún no hay comentarios